Search the Knowledge Hub Archive
SAGES Conference 2026: Connect to purpose
After attending the Society of American Gastrointestinal and Endoscopic Surgeons Conference up the road in sunny Tampa, FL, it was easy to see why this year’s theme, connect to purpose, was so fitting. Across lectures, panels, posters, and exhibits, a consistent narrative emerged: surgeons, KOLs, engineers, and medical device manufacturers are deeply aligned in a shared mission to improve patient outcomes and elevate provider experience.
Cutting through the noise in Precision Diagnostics: How CGP labs can better communicate & engage customers across the full offering
Tuesday 12 May, 2026 - 10:00 EDT | 15:00 BST | 16:00 CEST. In this complimentary 30-minute webinar, our Precision Medicine and Diagnostics experts explore why true differentiation is often overlooked in real-world practice, and what diagnostic companies can do to ensure their tests and portfolios are clearly understood, valued and adopted.
ACC Conference 2026: Cardiology at a turning point
After attending the American College of Cardiology (ACC) Conference 2026, Molly Hines and Supreet Kaur reflected on their key takeaways from the event. At ACC, much of the discussion was shaped by how artificial intelligence is increasingly being embedded into clinical practice, workflows, and healthcare systems.
Why Primary Care Matters in MCED: Precision Medicine & Diagnostics Newsletter, Volume 3
In the third edition of our Precision Medicine & Diagnostics Newsletter, our team explores why primary care matters in Multi-Cancer Early Detection (MCED) and why its future hinges not just on innovation, but on integration.
RAPS Euro Convergence, Lisbon
We are delighted to be attending, sponsoring and presenting at the RAPS Euro Convergence from 5-8 April in Lisbon. Purdie Pascoe’s Post Market Survey experts Marcus Torr (Head of Post Market Surveys) Ellie Baker (Commercial Director) Alice Robertson (Research Manager) and Ruby Pogson Jones (Senior Research Executive ) are exhibiting on booth 31.
Cracking complex MedTech decisions: How SIMALTO reveals what truly drives stakeholder choice
In this webcast our specialists Sabera Hyderally, Head of US MedTech and Kristin Lovallo, Director, will share how this advanced methodology can uncover deeper insights, helping organizations prioritize product features, refine value propositions, and make more confident strategic decisions.
Why qualitative research matters in MedTech (and why it’s often undervalued)
Behavioural economics has long highlighted the importance of context. Data without context will only get you so far. MedTech companies need to understand why clinicians behave the way they do, how decisions actually get made inside hospitals, what ‘risk’ really means to different stakeholders and where friction exists within workflows, perceptions and emotions.
Early Cancer Detection - Inside the Consumer Conversation: Precision Medicine & Diagnostics Newsletter, Volume 2
In the second edition of our Precision Medicine & Diagnostics Newsletter, our team explores the rise of early cancer detection through the lens of the consumer, covering growing interest in MCED, the shift toward proactive and direct-to-consumer testing, and the key awareness, perception, and education challenges shaping adoption.
American College of Cardiology Conference 2026
We are delighted to be attending the ACC Annual Conference from March 28–30 in New Orleans, Louisiana. Senior Research Executives Molly Hines and Supreet Kaur will be representing Purdie Pascoe at the event.
The Society of American Gastrointestinal and Endoscopic Surgeons
We are delighted to be attending the SAGES Annual Conference from March 25–28 in Tampa, Florida. Kristin Lovallo, US Research Director, will be representing Purdie Pascoe at the event.
Insights from the 2026 TT Life Sciences MedTech Clinical Network Conference in Brussels
The 2026 MedTech Clinical Network Conference, hosted by TT Life Sciences in Brussels, brought together clinical leaders from across Europe at a pivotal time for the MedTech sector. Rather than simply revisiting regulatory updates, this year’s discussions focused on a broader challenge: how organisations can navigate uncertainty, strengthen clinical strategy, and generate evidence that delivers both regulatory assurance and commercial value.
How we used a staged qualitative approach to help inform the development of a portfolio of novel digital solutions for GI endotherapy
Find out how we used a staged qualitative approach to help our client understand the landscape of digital solutions in GI endotherapy with a focus on AI, data management (clinical and non-clinical), workflow, and supply management.
Beyond the booth: Turning MedTech conferences into strategic intelligence
It already feels like we’re flying through 2026, and the next wave of major MedTech conferences is coming into view fast. Calendars are filling, budgets are being committed, and plans are taking shape. The question is no longer whether to attend, but whether your conference research strategy is truly ready?
Competing in the MRD race: Precision Medicine and Diagnostics Newsletter, Volume 1
In the first edition of our Precision Medicine & Diagnostics Newsletter, our dedicated team explores the evolving MRD landscape, highlighting increasing competition, the shift from technical performance to user experience, and the ongoing challenges around clinician confidence and consistent adoption.
The Global Shift in Real-World Evidence: Insights from MedTech Industry Leaders
Real-World Evidence (RWE) is playing an increasingly important role in global MedTech regulatory decision-making, supporting evidence generation across the product lifecycle, from pre-market submissions to post-market surveillance. Watch this webcast which features industry experts to understand how MedTech manufacturers can build effective, compliant RWE strategies.
New syndicated reports: Cancer monitoring US landscape - 4th wave now available
As the ctDNA cancer monitoring market becomes increasingly competitive, established panels continue to maintain their foothold amidst a surge of new entrants. Purdie Pascoe is conducting an ongoing syndicated study exploring the US ctDNA Cancer Monitoring Landscape. Waves 1 through 4 are now available for immediate purchase.
Purdie Pascoe’s Primary Market Research team
Our primary market research team combines deep expertise with a full range of qualitative and quantitative techniques, including the latest digital approaches with a wealth of experience across medical devices, digital healthcare, drug delivery, precision medicine and diagnostics.
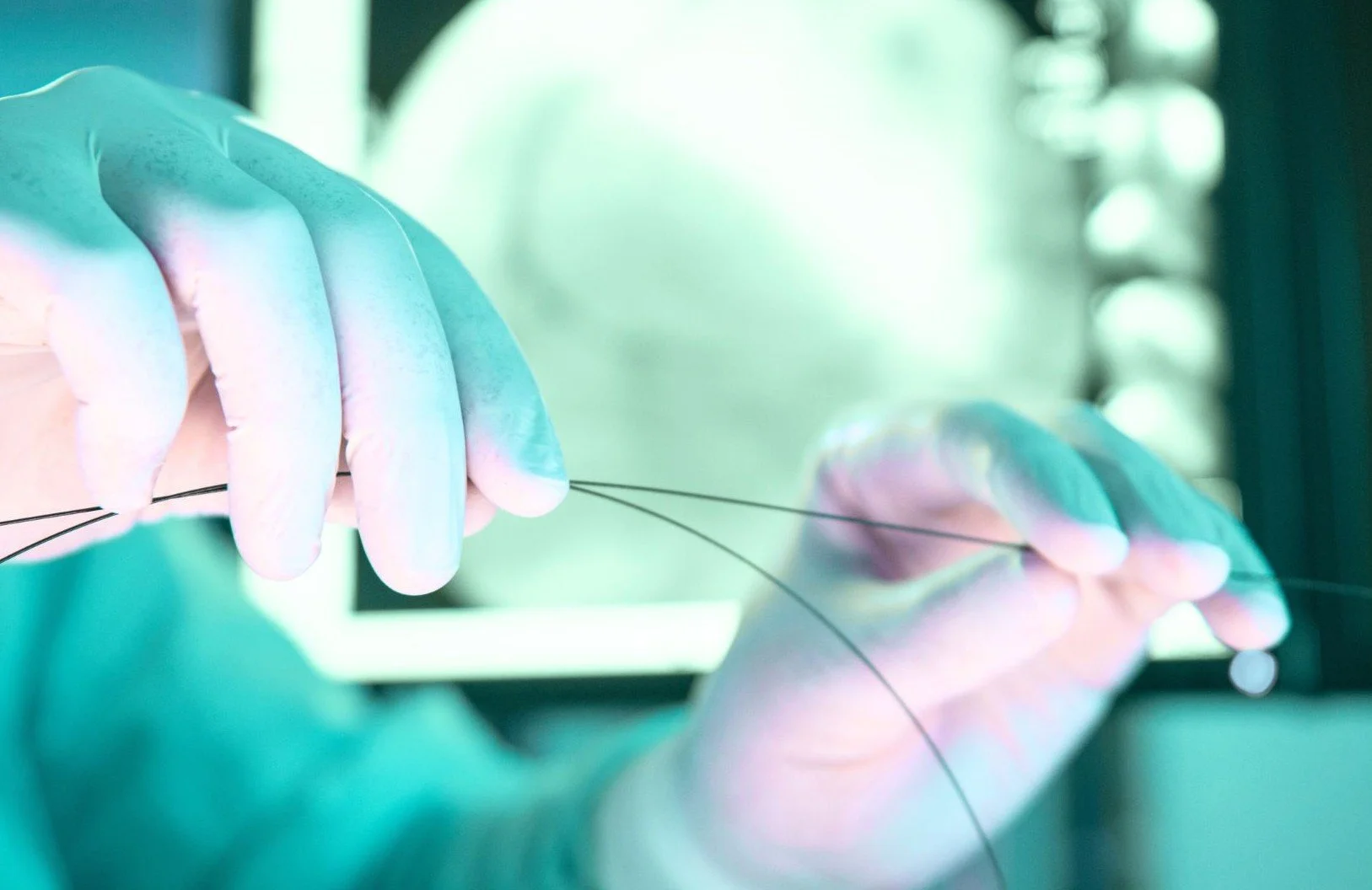
From data to confidence: PMCF survey success for a Class III Guidewire
As part of compliance with the EU Medical Device Regulation (MDR), the manufacturer of a Class III guidewire needed to generate robust Post Market Clinical Follow-Up (PMCF) data to confirm ongoing safety and performance. The study was designed to evaluate two key endpoints: procedural success, and freedom from adverse events.
Smarter workflows, sharper insights: How our dedicated Innovation team helps us (and our clients) work better
Innovation sits right at the intersection of data, technology, and research. Our Innovation team’s goal is simple: make complex studies easier to run, insights easier to find, and decisions easier to make, for both our research colleagues and our clients.
Real-World Data: The fuel behind modern clinical evidence, and why Post Market Surveys are a smart way to capture it
As regulators worldwide including the FDA and EU Notified Bodies place greater emphasis on RWD, the industry faces a familiar but intensified challenge: how to generate high-quality, device-specific evidence efficiently, reliably, and without escalating costs. That’s where Post-Market Surveys come in.